
One power unit operates a choice of support surfaces
Mix and match the PULSAIR Choice power unit with the mattress replacement (1-in-4 active cell cycle), the mattress overlay (1-in-2 active cell cycle) or the cushion (1-in-2 active cell cycle) for simple, cost-effective stepping up or down of pressure care for patients at all risks of pressure damage.

Mattress Replacement system with active 1-in-4 cycle and TISSUEgardTM
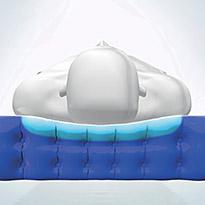
An active 1-in-4 cell cycle supports 75% of the patient’s body at all times, on groups of three inflated cells, whilst the fourth cell deflates sufficiently to redistribute the pressure and encourage tissue reperfusion. TISSUEgard enables the partial immersion and envelopment of the patient into the support surface, reducing the pressure on the patient’s skin and decreasing the pressure differential between inflated and deflated cells, which helps reduce tissue strain and associated shearing forces.

Mattress Overlay system with active 1-in-2 cycle
An active 1-in-2 cell cycle supports 50% of the patient’s body at all times on alternate inflated cells, whilst the other cells deflate sufficiently to redistribute the pressure and encourage tissue reperfusion.
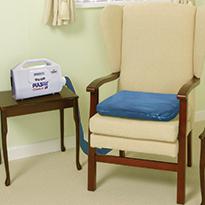
Cushion system with active 1-in-2 cycle
The PULSAIR Choice cushion system features a 1-in-2 cell cycle giving active pressure relief to the seated patient
Ortho-differential SupportTM
ODS results in firmer outer edges of the mattress which facilitate patient transfers and provide extra support, safety and comfort for larger or heavier patients. It also creates a softer central area of the mattress, ideal for smaller, lighter patients.

Multi-stretch cover
The Multi-stretch cover promotes tissue offloading during cell deflation and reduces shear and friction during patient movement or manual repositioning. The material is waterproof, moisture-vapour permeable and all seams are welded to protect the inside of the mattress from fluid ingress.
Microclimate management
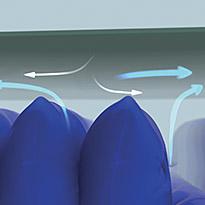
The waterproof, moisture-vapour permeable properties of the multi-stretch cover, combined with a low-air-loss feature within the mattress, helps manage the skin 'microclimate' between the patient and support surface.

Simply designed interface
The simply designed interface and easy-touch buttons allow the carer to control all essential functions such as the Soft, Medium or Firm comfort settings.

Patient Transport Facility
The Patient Transport Facility turns the mattress into a static unpowered support surface for up to 4 hours.

Fast, safe and easy manual interventions
Manual interventions such as CPR are fast, safe and easy to operate.




-A-DHG-Company-01-7-1614716979.png)

